New PET Imaging Study Reveals How Ketamine Relieves Treatment-Resistant Depression
2026.03.06
Researchers visualize molecular brain changes that explain ketamine’s rapid antidepressant action
Researcher(s): Takuya Takahashi, Waki Nakajima
Researchers from Japan have uncovered direct evidence showing how ketamine exerts its rapid antidepressant effects in patients with treatment-resistant depression, using a novel brain imaging technique to visualize molecular changes in the living human brain. Using positron emission tomography imaging with the newly developed tracer [¹¹C]K-2, the study shows that ketamine’s antidepressant effects are mediated by region-specific changes in AMPA receptor density that correlate with symptom improvement, bridging findings from animal models to human patients.
Major depressive disorder (MDD) is one of the leading causes of disability worldwide, and approximately 30% of patients develop treatment-resistant depression (TRD), a condition that does not respond adequately to conventional antidepressant therapies. Although ketamine has emerged as a rapid-acting antidepressant for individuals with TRD, its underlying biological mechanism in the human brain has remained poorly understood, limiting efforts to optimize and personalize treatment.
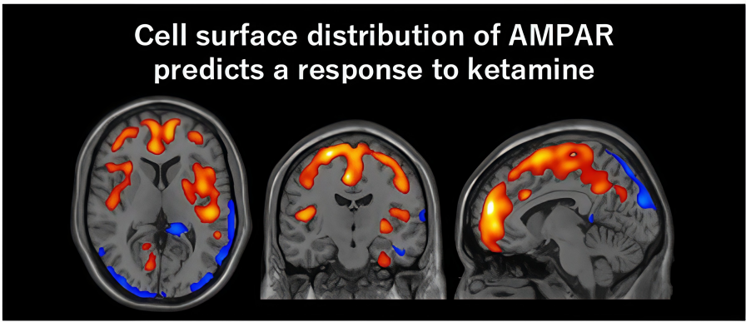
In a new study published in the journal Molecular Psychiatry on March 05, 2026, a research team led by Professor Takuya Takahashi from the Department of Physiology, Yokohama City University Graduate School of Medicine, Japan, employed an innovative positron emission tomography (PET) imaging approach to directly examine changes in glutamate α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor (AMPAR), a key protein involved in synaptic plasticity and glutamatergic signaling, in patients receiving ketamine. Prof. Takahashi noted, “Although ketamine has shown rapid antidepressant effects in patients with treatment-resistant depression, its molecular mechanism in the human brain has remained unclear.”
This advance was made possible by the team’s previously developed PET tracer, [¹¹C]K-2, which enables visualization of cell-surface AMPAR in the living human brain. While preclinical studies have long suggested that ketamine’s antidepressant effects depend on AMPAR activity, this study provides the first direct evidence supporting this mechanism in humans.
The study integrated data from three registered clinical trials conducted in Japan and included 34 patients with TRD and 49 healthy control participants. Patients received intravenous ketamine or a placebo over a two-week period, with PET imaging performed before treatment initiation and after the final infusion.
Results revealed that individuals with TRD exhibited widespread, region-specific abnormalities in AMPAR density compared with healthy participants. Notably, ketamine did not induce uniform changes across the brain. Instead, clinical improvement was associated with dynamic, region-specific modulation of AMPAR. Increases in receptor density were observed in several cortical regions, while decreases were detected in reward-related areas, particularly the habenula. These region-specific changes were strongly correlated with reductions in depressive symptoms.
“Ketamine’s antidepressant effect in patients with TRD is mediated by dynamic changes in AMPAR in the living human brain,” Prof. Takahashi explained. “Using a novel PET tracer, [¹¹C]K-2, we were able to visualize how ketamine alters AMPAR distribution across specific brain regions and how these changes correlate with improvements in depressive symptoms.” These findings provide direct human evidence linking molecular mechanisms previously identified in animal models to clinical antidepressant effects.
Beyond advancing mechanistic understanding, the findings have important clinical implications. AMPAR PET imaging may represent a valuable biomarker for evaluating and predicting individual response to ketamine treatment in TRD. Given the substantial proportion of patients who do not benefit from standard antidepressants, the identification of such biomarkers addresses a critical unmet need in mental healthcare.
By directly visualizing AMPAR dynamics in the living human brain, this study bridges a longstanding gap between preclinical research and clinical psychiatry. The results establish AMPAR modulation as a central molecular mechanism underlying ketamine’s rapid antidepressant effects and highlight AMPAR PET imaging as a promising tool for guiding personalized treatment strategies. Ultimately, this work may accelerate the development of more precise, targeted therapies for individuals with treatment-resistant depression.
In a new study published in the journal Molecular Psychiatry on March 05, 2026, a research team led by Professor Takuya Takahashi from the Department of Physiology, Yokohama City University Graduate School of Medicine, Japan, employed an innovative positron emission tomography (PET) imaging approach to directly examine changes in glutamate α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor (AMPAR), a key protein involved in synaptic plasticity and glutamatergic signaling, in patients receiving ketamine. Prof. Takahashi noted, “Although ketamine has shown rapid antidepressant effects in patients with treatment-resistant depression, its molecular mechanism in the human brain has remained unclear.”
This advance was made possible by the team’s previously developed PET tracer, [¹¹C]K-2, which enables visualization of cell-surface AMPAR in the living human brain. While preclinical studies have long suggested that ketamine’s antidepressant effects depend on AMPAR activity, this study provides the first direct evidence supporting this mechanism in humans.
The study integrated data from three registered clinical trials conducted in Japan and included 34 patients with TRD and 49 healthy control participants. Patients received intravenous ketamine or a placebo over a two-week period, with PET imaging performed before treatment initiation and after the final infusion.
Results revealed that individuals with TRD exhibited widespread, region-specific abnormalities in AMPAR density compared with healthy participants. Notably, ketamine did not induce uniform changes across the brain. Instead, clinical improvement was associated with dynamic, region-specific modulation of AMPAR. Increases in receptor density were observed in several cortical regions, while decreases were detected in reward-related areas, particularly the habenula. These region-specific changes were strongly correlated with reductions in depressive symptoms.
“Ketamine’s antidepressant effect in patients with TRD is mediated by dynamic changes in AMPAR in the living human brain,” Prof. Takahashi explained. “Using a novel PET tracer, [¹¹C]K-2, we were able to visualize how ketamine alters AMPAR distribution across specific brain regions and how these changes correlate with improvements in depressive symptoms.” These findings provide direct human evidence linking molecular mechanisms previously identified in animal models to clinical antidepressant effects.
Beyond advancing mechanistic understanding, the findings have important clinical implications. AMPAR PET imaging may represent a valuable biomarker for evaluating and predicting individual response to ketamine treatment in TRD. Given the substantial proportion of patients who do not benefit from standard antidepressants, the identification of such biomarkers addresses a critical unmet need in mental healthcare.
By directly visualizing AMPAR dynamics in the living human brain, this study bridges a longstanding gap between preclinical research and clinical psychiatry. The results establish AMPAR modulation as a central molecular mechanism underlying ketamine’s rapid antidepressant effects and highlight AMPAR PET imaging as a promising tool for guiding personalized treatment strategies. Ultimately, this work may accelerate the development of more precise, targeted therapies for individuals with treatment-resistant depression.

Usage restrictions: Credit must be given to the creator.
Reference
Title of original paper: The dynamics of AMPA receptors underlies the efficacy of ketamine in treatment resistant patients with depression
Journal: Molecular Psychiatry
DOI: 10.0.4.14/s41380-026-03510-w
Journal: Molecular Psychiatry
DOI: 10.0.4.14/s41380-026-03510-w
Additional information
Latest Article Publication Date: 5 March 2026
Conflicts of Interest Statement: Takuya Takahashi is the inventor of a patent application for a novel compound that specifically binds to the AMPA receptor (WO 2017006931), including [11C]K-2. Takuya Takahashi and Tetsu Arisawa are the founders and stockholders of AMPAMETRY, Inc., which holds the exclusive license to use [11C]K-2. No other potential conflicts of interest relevant to this article exist.
Conflicts of Interest Statement: Takuya Takahashi is the inventor of a patent application for a novel compound that specifically binds to the AMPA receptor (WO 2017006931), including [11C]K-2. Takuya Takahashi and Tetsu Arisawa are the founders and stockholders of AMPAMETRY, Inc., which holds the exclusive license to use [11C]K-2. No other potential conflicts of interest relevant to this article exist.
About Professor Takuya Takahashi from Yokohama City University Graduate School of Medicine, Japan
Dr. Takuya Takahashi is a Professor at the Yokohama City University Graduate School of Medicine, Japan. His research group primarily studies AMPA receptors, plasticity, synapses, stress hormones, and the comprehensive brain networks. He has over 120 publications to his credit and received several research awards, including the Yokohama City University’s Chairman Award, National Alliance for Research’s Young Investigator Award, Tsukahara Nakaakira Memorial Award, and the Commendation for Science and Technology by the Ministry of Education, Culture, Sports, Science and Technology (MEXT). Prof. Takahashi is a highly cited researcher, with work published in reputed journals such as Science, Cell, Scientific Reports, and Nature Medicine.Funding information
This work was supported by the Ministry of Education, Culture, Sports, Science and Technology (Special Coordination Funds for Promoting Science and Technology); the Japan Agency for Medical Research and Development (AMED) (grant numbers: JP18dm0207023, JP19dm0207072, JP24wm0625304, JP25gm7010019, and JP20dm0107124); the Japan Society for the Promotion of Science KAKENHI (grant numbers: 22H03001, 20H00549, 20H05922, 23K10432, 19H03587, 20K20603, 22K15793, and 21K07508); the Takeda Science Foundation; the Keio Next-Generation Research Project Program; the SENSHIN Medical Research Foundation; and the Japan Research Foundation for Clinical Pharmacology.For inquiries regarding this press release
Takuya Takahashi
Professor
Department of Physiology, Graduate School of Medicine
Yokohama City University
Professor
Department of Physiology, Graduate School of Medicine
Yokohama City University
Media contact
Yokohama City University, Public Relations Division
koho@yokohama-cu.ac.jp
koho@yokohama-cu.ac.jp
